|
4/19/2023 0 Comments Atomic radius trend
Join my 10,000+ subscribers on my YouTube Channel for new A Level Chemistry video lessons every week. Please LIKE this video and SHARE it with your friends! Topic: Periodicity, Inorganic Chemistry, A Level Chemistry, Singaporeīack to other previous Inorganic Chemistry Video Lessons.įound this A Level Chemistry video useful? Overall the effective nuclear charge increases, there is a stronger attraction between nucleus and valence electrons, the nucleus can pull the electrons closer to itself, hence ionic radius decreases. Within the cations and anions, the explanation will go back to effective nuclear charge.Īll cations (and anions) are isoelectronic with the same number of electrons and electronic configuration, hence shielding effect is the same. Therefore all anions with 3 electron shells will be larger than all cations with only 2 electron shells. The next 3 elements (P, S, Cl) gain electrons to form anions with 18 electrons, hence will fill up 3 principal quantum shells. The first 4 elements (Na, Mg, Al, Si) lose electrons to form cations with 10 electrons, hence will fill up 2 principal quantum shells. Decrease in radii within cations and anions 
There is a clear trend in atomic radius when going down the elements in a. There are 2 observations for ionic radii:ī. Atomic radius measures the distance between the nucleus and the outermost electron(s). Generally, there is a decrease in the atomic radius while moving from left to right in a period, while there is an increase in the same while moving down a group. This trend can be explained by considering the energy level and the nuclear charge. Overall the effective nuclear charge increases, there is a stronger attraction between nucleus and valence electrons, the nucleus can pull the electrons closer to itself, hence atomic radius decreases. The variation of the atomic radius in the periodic table follows a fixed pattern. Note that effective nuclear charge is also used to explain first ionisation energy trend across Period 3.Īcross Period 3, proton number increases, the nucleus is more positively charged hence nuclear charge increases.Įlectrons are added to the same principal quantum shell, do not shield each other since they are the same distance from the nucleus, hence shielding effect is the same. We can use the concept of effective nuclear charge (ENC) to explain the atomic radii. The atomic radii decreases across Period 3. 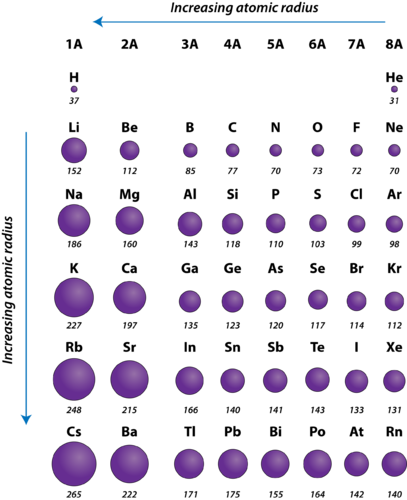
In this video we want to dicuss the atomic and ionic radii of Period 3 elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
